Acid-Base Imbalance
Acid-base imbalance is an abnormality normal balance of acids and
bases of the human body's that causes the deviation of plasma
pH from normal range (7.35 - 7.45).
Know the role of buffers in acid-base balance.
Acid-base Disorders Result From-
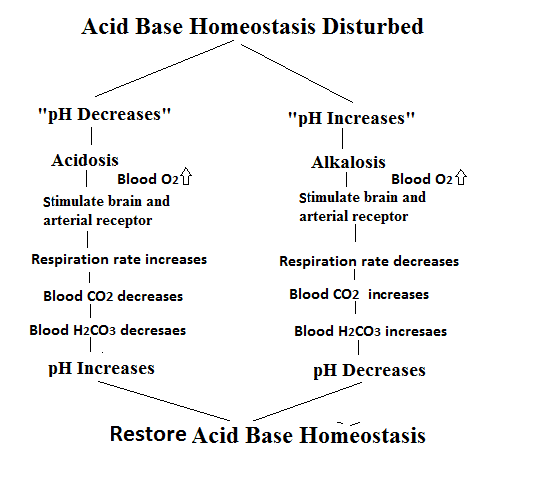
1. Acidosis
When pH is lesser than normal range, condition is
called Acidosis.
Acidosis is a condition in which
there is build-up too much acid in the body
fluids.
Acidosis condition states build-up of acid in blood stream. Acid accumulates and bases are lost.
Acidosis is the opposite of
alkalosis (a condition in which excessive base in the body fluids).
2. Alkalosis
When pH is more than normal range, condition is called
Alkalosis.
Alkalosis condition refers excessive blood alkalinity
caused by overabundance of bicarbonate in blood or loss of acid from blood.
In this state base accumulates or acid is lost.
Alteration in Acid-Base Balances
Metabolic Causes are Responsible for –
1. Metabolic acidosis
2. Metabolic alkalosis
Respiratory Causes Result in –
1. Respiratory acidosis
2. Respiratory alkalosis
1. Metabolic Acidosis
Metabolic acidosis is most common acid-base disturbance in clinical
practice.
Metabolic acidosis has three
main root causes –
1. Increased acid production and loss of sodium
bicarbonate.
2. Concentration
of HCO3 ion is reduced due to increased production of acids.
3. Reduce ability of kidney to excrete excess Acids.
Know the role of the kidney in acid-base balance.
This condition compensated by respiratory system
through lowering pCO2 and carbonic acid concentration in blood or through
renal compensation by increasing excretion of H+ in urine.
Causes of Metabolic Acidosis are -
1. Uncontrolled diabetes mellitus
2. Kidney failure
3. Diarrhoea
4. Vigorous exercise
5. Salicylate
toxicity, metformin toxicity, sulfate
toxicity
6. Organic acidemias (defect in protein metabolism; essential
enzyme is absent or malfunctioning). This disorders characterized by the excretion of organic acids in the
urine.
2. Metabolic Alkalosis
Metabolic alkalosis occurs due to Increased HCO3- concentration in plasma which
raises pH and abnormal loss of acid.
This condition result of decreased H+ ion concentration,
and increased bicarbonate concentration.
Metabolic alkalosis compensated by rise in pCO2 by
hypoventilation following increased excretion of HCO3- in
urine.
Metabolic Alkalosis Occur in -
1. Prolonged vomiting
2. Gastric lavage - clearing out content of stomach
3. Bicarbonate ingestion in peptic ulcer
4. Urea cycle defects
3. Respiratory Acidosis
Respiratory acidosis is condition occurs
when high levels of acid in the blood due to increased levels of carbon dioxide
(CO2) in the body, results
in retention of CO2 and fall in blood pH.
Arise due to hypoventilation, excess CO2 causes the decrease in pH of blood, making them too acidic.
Inadequate Ventilation Occurs due to
Obstruction of Respiration, seen in -
1. Pneumonia
2. Asthma
3.
Emphysema
4. Respiratory center depression
5. Alcohol
ingestion
4. Respiratory Alkalosis
Respiratory Alkalosis caused by hyperventilation that leads to decrease concentration of CO2. Decreased pCO2 results in less production of H+ and rise in pH of blood plasma.
Respiratory Alkalosis Occurs due to-
1. Anxiety
2. CNS injury
3. Fever
4. Lack of oxygen at high altitude
5. Hot bath, increased environmental
temperature
To overcome this condition Kidneys compensate for respiratory alkalosis by decreased H+ secretion and reduced reabsorption of HCO3-.
Diagnostic Tests for Acid- Base Disorders
1. Arterial
Blood Gases (ABG) Analysis
Analysis of blood gases pO2, pCO2, O2, HCO3, and pH level
Blood sample taken from artery (femoral
artery of groin, brachial artery in the arm or radial artery in wrist)
2. Serum electrolytes
Measure level of body’s main electrolytes.
3. Bicarbonate ion estimation
4. Anion gap
5. Serum creatinine, Blood urea
estimation, and Urine PH








0 Comments