Acid-Base Balance
Normal pH range of blood 7.35 to 7.45. To evaluate the acid-base
balance of a person is by measuring pH and levels of carbon dioxide (acid) and
bicarbonate (base) in blood.
The alkalinity of blood increases when the level of base
increases or when the level of acid in the body decreases.
Know here the pH buffer systems of the body.
1. Acid
Acid is a substance, ion, or molecule that librates
(free) H+ ions when dissolving in water. Their pH values of
acid are always less than 7. Acids are sour-tasting substances.
HCL - H+ + Cl-
Acid is weak or strong depending upon the percentage
availability of H+ ions.
For example
- Strong Acid; Hydrochloric acid
Weak Acid; Acetic acid
The addition of acid to the solution increases the concentration
of free H+ ions, which makes the solution more acidic, resulting in a decrease in pH (Power
of hydrogen).
2. Base
The base is a substance, ion, or molecule that accepts H+
ions. Base substances release hydroxide ions (OH– ions) when
dissolved in water (aqueous solution). The pH
values of the base are
always greater than 7. Bases are bitter-tasting substances.
These OH– ion combines with free H+ ions and thus lowers H+ ion
concentration,
resulting in an increase in pH of a solution.
For example - Sodium hydroxide
NaOH - Na+ + OH–
There are two types of litmus paper; Red litmus paper and blue litmus
paper used to identify acids and bases –
1. Under acidic conditions, Blue litmus paper turns red and,
2. Under basic or alkaline conditions Red litmus paper turns blue.
3. Amphoteric Substance
A substance that are able to act both as acid and as
well as base or Molecule, an ion that can react both as an acid and as a base.
Examples
of Amphoteric substances include; water,
proteins, amino acids, and many metal oxides and hydroxides.
Water is the most common
amphoteric substance which can act
either as Acid or Base.
4. Neutral Substance
A
neutral substance is a substance that has no acid or base properties, has the same
amount of hydrogen and hydroxyl ions.
Neutral substances have a pH of 7.0 is
neutral.
For example-
pH value of pure water is 7.
pH value of common salt (sodium chloride) is about equal to 7.
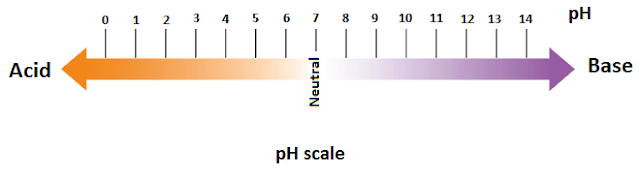
5. PH (Power of Hydrogen)
It is a measure of (H+) ion actively in the water (aqueous solution).
PH expressed on pH scale from 0 to 14. pH with 7 is neutral, representing
neutrality. pH of less
than 7 indicates an acid, and pH of greater than 7 indicates a base.
pH of Different Biological fluids is -
pH of Saliva
- Normal Saliva pH range is 6.2-7.6, average pH is 6.7
pH of Urine
-Normal Urine pH range is between 4.5 – 8, average pH is
6.0
pH of Blood -
Normal Blood pH range from 7.35 - 7.45
pH of Gastric fluid - Normal Blood pH range from1.5 to 3.5
Check out here what are the types of acid-base imbalance?
Importance of pH
1. pH can be affected by chemical in the water, pH is an
important indicator of water of what changing chemically.
2. To measures acidic and basic condition of blood.
3. To measure availability of essential nutrient.
4. To control the biological functions, behaviour of chemicals and, microbial
activity.








0 Comments